In the rapidly evolving landscape of clinical trials, two terms that are gaining significant traction are “digital endpoints” and “patient outcomes.” Both play critical roles in the assessment of new medical interventions, yet they serve distinct purposes and offer unique insights into the effectiveness and impact of treatments. Understanding the differences and similarities between these two components is essential for researchers, clinicians, and patients alike. In this blog, we’ll explore what digital endpoints and patient outcomes entail, their value in clinical trials, and provide examples to illustrate their application.
Digital Endpoints: The New Frontier in Clinical Data
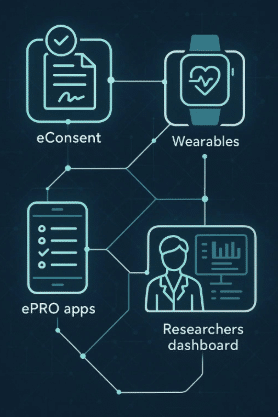
Digital endpoints refer to measures obtained through digital devices such as wearables, smartphones, and other sensor-based technologies. These endpoints can capture a wide range of data including physical activity, sleep patterns, heart rate, and more. For example, a smartwatch might be used to continuously monitor a patient’s heart rate variability, which could serve as a digital endpoint in a study evaluating a new cardiovascular drug.
The value of digital endpoints lies in their ability to provide continuous, objective, and high-resolution data. Unlike traditional measures, which might only capture a snapshot of a patient’s health at a specific time, digital endpoints can offer a more comprehensive and real-time picture of the patient’s condition. This can lead to more nuanced and dynamic understanding of treatment effects.
Patient Outcomes: The Heart of Clinical Trials
Patient outcomes, on the other hand, refer to the impacts of medical interventions on the health and quality of life of patients. These outcomes can be subjective, such as self-reported pain levels, or objective, like survival rates or hospital readmission rates. Patient outcomes remain the gold standard for assessing the efficacy of treatments, as they directly reflect the benefits (or harms) experienced by patients.
The value of patient outcomes is evident: they provide a clear and direct measure of what matters most to patients. Whether a new treatment improves life expectancy, reduces symptoms, or enhances quality of life, patient outcomes help determine the real-world value of medical interventions.
Bridging the Gap: Similarities and Integration
While digital endpoints and patient outcomes may seem different at first glance, they share the common goal of improving patient care. Both are crucial for understanding the effectiveness of treatments and guiding clinical decision-making. Moreover, digital endpoints can complement traditional patient outcomes by providing additional context and insights into patient health.
For instance, in a study on a new insomnia medication, traditional patient-reported outcomes might include self-assessed sleep quality or time taken to fall asleep. A corresponding digital endpoint could be data collected via a wearable device tracking actual sleep patterns and nocturnal activity. Together, these measures can provide a more complete picture of the treatment’s effectiveness.
Examples in Clinical Trials
Consider a clinical trial for a new diabetes medication. Traditional patient outcomes might include HbA1c levels (a measure of blood sugar control), incidence of diabetes-related complications, and patient-reported quality of life. A digital endpoint in the same trial could be continuous glucose monitoring using a digital device, providing detailed insights into daily blood sugar fluctuations.
Another example is in the context of Parkinson’s disease research. Patient outcomes might focus on motor function and quality of life assessments. Digital endpoints could include sensor-based measurements of tremor frequency and amplitude, offering objective data to complement patient reports.
The Future of Clinical Trials
The integration of digital endpoints with traditional patient outcomes represents the future of clinical trials. This approach allows for a more holistic view of treatment effects, combining objective data with patient-centered outcomes. As technology advances, the use of digital endpoints is likely to increase, enhancing our ability to assess and understand the full impact of medical interventions.
In conclusion, while digital endpoints and patient outcomes serve different functions in clinical trials, they are not mutually exclusive. Instead, they are complementary tools that, when used together, can provide a richer and more accurate picture of a treatment’s effectiveness. As we move forward, the harmonious integration of these elements will be pivotal in driving advancements in medical research and patient care.
The Combined Value in Clinical Research
By leveraging both digital endpoints and patient outcomes, clinical trials can achieve a holistic view of treatment impacts. This approach not only enhances the understanding of disease patterns and treatment effects but also aligns medical research more closely with patients’ needs and experiences.
As technology advances and patient-centered care becomes increasingly important, the integration of digital endpoints and patient outcomes will likely play a pivotal role in the future of clinical trials. This synergy not only advances scientific knowledge but also ensures that patient care remains at the heart of medical research.
References
For those interested in delving deeper into this topic, I recommend exploring articles such as:
- “Digital Endpoints in Parkinson’s Disease: An Unbiased Approach to Disease Modelling” published in the Journal of Parkinson’s Disease.
- “The STAR*D Project Results: A Comprehensive Review of Findings” available in the American Journal of Psychiatry.
These articles provide detailed insights into the use of digital endpoints and patient outcomes in clinical trials, offering a glimpse into the future of medical research.