Introduction: Compliance + Retention = ROI That Matters
The clinical research landscape is rapidly transforming. Sponsors and CROs face mounting pressure to deliver trials that are faster, more cost-effective, and patient-centric. Yet the greatest threat to trial success isn’t recruitment — it’s retention and compliance.
Every patient who drops out creates ripple effects:
- Higher cost per retained participant.
- Increased recruitment expenses.
- Delayed database lock and submission timelines.
- Millions lost in delayed revenue opportunities.
That’s why ePRO (electronic Patient-Reported Outcomes) and eCOA (electronic Clinical Outcome Assessments) are no longer “nice to have.” They are critical ROI drivers. By improving compliance, reducing dropouts, and generating higher-quality data, they directly accelerate regulatory submission and time-to-market.
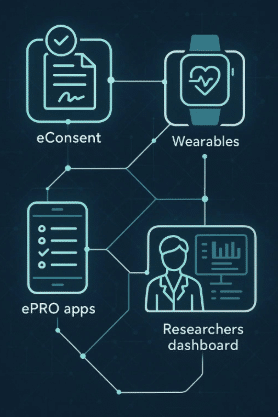
Delve Health’s Clinical StudyPal platform goes further. By unifying ePRO/eCOA, wearables, eConsent, and multilingual concierge services, it delivers both operational ROI (cost savings, efficiency gains) and strategic ROI (faster approvals, earlier revenue, competitive advantage).
What ROI Means in Clinical Trials
ROI in clinical trials is broader than traditional financial return. It includes:
- Compliance & Retention Gains → more complete datasets and fewer costly re-recruits.
- Operational Efficiency → automation and streamlined workflows.
- Data Quality → fewer queries, better regulatory readiness.
- Time-to-Market Acceleration → earlier FDA/EMA submission, faster commercial revenue.
- Long-Term Strategic Advantage → future-proofing trials for decentralized and hybrid models.
5 ROI Drivers with Delve Health’s Integrated Platform
1. Compliance & Retention: The Core of ROI
Patient compliance and retention are the #1 predictors of trial success. ePRO/eCOA systems support engagement by:
- Enabling mobile-first reporting with reminders and notifications.
- Using wearables to passively capture vitals and activity data.
- Providing multilingual concierge services to resolve technical, scheduling, or travel issues in real time.
📈 Impact: Delve Health clients have seen 15–30% improvements in retention, reducing costly dropout replacement and accelerating trial timelines.
2. Efficiency Gains That Shorten Timelines
Digitizing outcomes dramatically reduces site and staff burden.
- Automated data capture replaces transcription and manual cleaning.
- Real-time dashboards identify compliance issues early.
- Adaptive trial readiness supports faster protocol adjustments.
📈 Impact: Sponsors achieve database lock up to 6–8 weeks faster, directly enabling earlier FDA/EMA submission.
3. Data Quality & Regulatory Confidence
High-quality data is the backbone of approvals. Delve Health’s platform ensures:
- Built-in validations and skip logic reduce errors and queries.
- Wearables add continuous, objective evidence.
- Audit-ready compliance with 21 CFR Part 11, GDPR, HIPAA, and GCP.
📈 Impact: Cleaner datasets reduce regulatory delays, resubmissions, and inspection risk — shortening approval timelines.
4. Resource Optimization & Vendor Consolidation
Traditional CRO setups often require multiple vendors for eCOA, eConsent, and wearable integration. This drives costs and slows workflows.
Delve Health simplifies trial operations with a single integrated platform.
- Sites spend less time chasing patients.
- Sponsors avoid duplicate vendor costs.
- Multilingual engagement scales globally.
📈 Impact: Significant cost reduction + faster study execution = stronger ROI.
5. Strategic Value: From Patients to Markets
Delve Health’s platform isn’t just about today’s trial — it future-proofs for tomorrow.
- Seamless support for decentralized, hybrid, and global trials.
- Integration of digital biomarkers for emerging therapeutic endpoints.
- Shared dashboards for stakeholder alignment.
📈 Impact: Drugs and devices reach patients months sooner, avoiding $30M–$300M per month in lost market opportunity.
Regulatory Alignment: ROI Through Risk Reduction
Regulators are raising expectations around digital data capture:
- FDA’s 2023 draft guidance on Digital Health Technologies emphasizes validated electronic data capture and remote monitoring.
- EMA reflections highlight the need for harmonized electronic systems for global trials.
- ICH E6(R3) GCP draft underscores digital transparency and traceability.
📈 Impact: ePRO/eCOA platforms like Delve Health’s don’t just improve efficiency — they de-risk trials by aligning with evolving regulatory frameworks.
Case Study: ROI in Action
In a Phase III oncology trial with 250 patients:
- Delve Health’s Clinical StudyPal improved retention by 22%.
- Protocol deviations dropped by 18%.
- Time-to-database-lock was reduced by 6 weeks.
💰 Estimated ROI: The accelerated submission translated into $90M in earlier market revenue for the sponsor — plus reduced re-recruitment costs.
Financial Math: Quantifying ROI
- Recruitment costs average $6,500–$12,000 per patient in Phase II/III studies. Improving retention by just 10% in a 500-patient trial saves $325,000–$600,000 in recruitment costs alone.
- Each month of approval delay costs sponsors $30M–$300M in lost commercial opportunity, depending on therapeutic area.
- Delve Health’s 15–30% retention boost + faster database lock = tens of millions in value creation per trial.
Competitive Differentiation: Why Delve Health Wins
Challenge | Traditional CRO / eCOA Vendor | Delve Health |
Retention & Compliance | Limited, site-led | Multilingual concierge + automation = 15–30% higher retention |
Vendor Complexity | Multiple vendors for eCOA, eConsent, wearables | One integrated platform |
Data Capture | Siloed systems | Unified with real-time dashboards |
Global Scale | Limited multilingual support | 65+ languages supported |
Regulatory Alignment | Basic audit trails | 21 CFR Part 11, HIPAA, GDPR compliance built in |
Measuring ROI Effectively
- Establish Baseline Metrics → dropout %, query rates, time-to-lock.
- Track KPIs Aligned with Retention + Compliance → retention rate, protocol deviation reduction, PRO completion.
- Tie Operational Gains to Commercial Outcomes → translate faster lock into submission gains + revenue impact.
- Continuously Optimize → use insights to refine trial design and workflows.
Conclusion: ROI Powered by Compliance & Retention
ROI in clinical trials is no longer just about cutting costs. It’s about ensuring patients stay engaged, data stays compliant, and trials move faster toward approval.
Delve Health delivers ROI at every level:
- Compliance & Retention → fewer dropouts, stronger datasets.
- Efficiency & Data Quality → faster lock, fewer queries.
- Strategic Value → faster approvals, earlier revenue, patient-first reputation.
👉 Ready to improve retention, compliance, and ROI in your next study? [Contact Delve Health today] to see how our integrated ePRO/eCOA, eConsent, wearables, and concierge services accelerate trial success.
Expanded FAQs (SEO-Driven)
What is the biggest hidden cost of poor retention in clinical trials?
Every dropout requires replacement, driving up recruitment costs and delaying timelines. Lost patients can add millions in expenses and months of delay.
How does ePRO improve protocol compliance?
Real-time prompts and automated reminders ensure patients complete assessments on schedule, reducing missed visits and deviations.
What’s the ROI difference between hybrid and fully decentralized trials?
Hybrid trials benefit from reduced site visits and improved compliance, while fully decentralized trials maximize efficiency by removing physical barriers — both drive ROI through retention and faster timelines.
How do wearable devices reduce trial costs?
Wearables cut down on site visits, capture real-world evidence, and reduce the need for additional monitoring visits, directly saving operational costs.
Can ROI be measured in patient experience?
Yes. Higher patient satisfaction leads to better compliance, fewer dropouts, and improved reputation for future studies — all of which contribute to ROI.