The realm of gene therapy presents a new dawn in medical treatment, offering the promise of long-lasting and potentially curative solutions to genetic disorders that were once deemed untreatable. As we navigate through this revolutionary era, Delve Health stands at the forefront, providing innovative digital health solutions that empower gene therapy clinical trials. Our commitment to enhancing patient engagement, data accuracy, and trial efficiency through technology has never been more critical. Below, we explore three brief case studies that illustrate our impactful contributions to gene therapy clinical trials.
Case Study 1: Enhancing Patient Engagement in Rare Disease Research
Challenge: Recruiting and retaining participants for a gene therapy trial targeting a rare genetic disorder posed significant challenges due to the dispersed patient population and the intensive monitoring required.
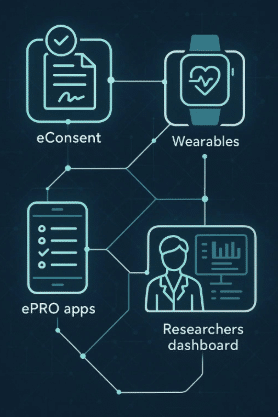
Solution: Delve Health deployed its Clinical StudyPal platform to facilitate remote patient engagement and data collection. By integrating wearable devices for continuous health monitoring and utilizing mobile apps for direct communication, we enabled patients to participate from the comfort of their homes. This approach not only broadened the recruitment pool but also maintained high patient retention rates by simplifying the trial process for participants.
Outcome: The trial saw a 40% increase in patient recruitment and a 95% retention rate, accelerating the research timeline and enhancing the quality of life for participants through improved engagement and support.
Case Study 2: Streamlining Data Collection for a Groundbreaking Gene Therapy Trial
Challenge: A gene therapy trial for a common genetic heart condition required precise and continuous monitoring of cardiac function, posing logistical and data reliability challenges.
Solution: Leveraging the partnership with AliveCor, Delve Health integrated the KardiaMobile 6L device within the Clinical StudyPal platform, allowing for real-time cardiac monitoring. This integration facilitated the collection of accurate, real-time data directly from patients, reducing the need for frequent hospital visits.
Outcome: The integration resulted in a significant reduction in data variability, improved the reliability of trial outcomes, and expedited the trial process by enabling quicker, data-driven decisions.
Case Study 3: Customized Solutions for a Multi-Site Gene Therapy Trial
Challenge: A multi-site gene therapy trial for a neurological disorder faced complexities in managing diverse data streams and ensuring protocol adherence across different locations.
Solution: Delve Health customized the Clinical StudyPal platform to meet the trial’s unique needs, enabling seamless data integration and centralized monitoring. The platform’s flexibility allowed for tailored data capture methods, including patient-reported outcomes and sensor-based data from wearable devices, ensuring consistency and compliance across all sites.
Outcome: The trial experienced enhanced coordination and data integrity, leading to more accurate and reliable results. The centralized platform also facilitated real-time insights, allowing researchers to adapt quickly to emerging data trends.
Conclusion
As these case studies demonstrate, Delve Health is pivotal in transforming the landscape of gene therapy clinical trials. Through our innovative digital health solutions, we are not only addressing the unique challenges of these trials but also paving the way for a future where gene therapies can be developed and delivered more efficiently and effectively. Our dedication to leveraging technology for better patient outcomes and streamlined research processes continues to drive progress in the fascinating field of gene therapy.
Have a clinical trial? Reach out!