In recent years, the healthcare industry has seen an exponential increase in the use of wearables for remote monitoring, both in standard healthcare settings and within clinical trials. These devices, which range from fitness trackers to specialized medical devices, offer unprecedented opportunities for continuous health data collection. However, as their use becomes more widespread, a significant challenge has emerged: wear compliance. Ensuring that patients consistently wear these devices and do so correctly has proven to be a multifaceted issue, impacting the reliability and efficacy of remote monitoring.
Understanding Wear Compliance
Wear compliance refers to the regular and proper use of wearable devices as prescribed by healthcare providers or clinical trial protocols. High compliance rates are crucial for gathering accurate and complete health data. However, factors such as discomfort, lack of motivation, privacy concerns, and technical difficulties can all contribute to non-compliance, leading to gaps in data and potentially compromising patient care and trial outcomes.
Identifying the Challenges
Several factors contribute to wear compliance issues:
Comfort and Convenience: If a wearable device is bulky, heavy, or uncomfortable, patients are less likely to wear it continuously. Comfort is paramount, especially for devices meant to be worn 24/7.
User-Friendliness: Devices that are complex to use or understand can deter patients, especially those who are not tech-savvy. Simplicity in design and function is essential.
Motivation and Engagement: Patients may lack the incentive to wear the device continuously, especially if they do not understand its importance or do not receive feedback about their data.
Privacy and Trust: Concerns about data privacy and security can make patients hesitant to use these devices, fearing misuse of their personal health information.
Technical Issues: Problems such as battery life, connectivity, and data accuracy can lead to frustration and discontinuation of use.
- Lifestyle Interference: Wearables that interfere with daily activities or are not waterproof can be significant obstacles.
- Forgetfulness: Simply forgetting to wear the device or charge it can lead to significant gaps in data.
These issues can severely impact the quality of data collected, leading to incomplete or inaccurate datasets that can compromise the outcomes of healthcare monitoring and clinical trials.
Clinical StudyPal: Managing Compliance Remotely
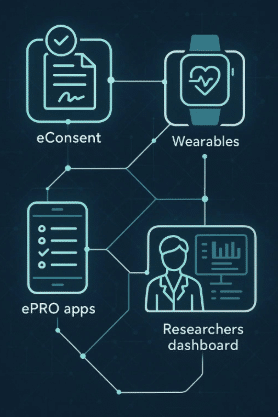
Clinical StudyPal is an innovative solution designed to address wear compliance issues effectively. It leverages technology and patient engagement strategies to ensure higher compliance rates, thus enhancing the quality and reliability of remote monitoring data. Here’s how Clinical StudyPal can help:
Customized Reminders and Notifications: Clinical StudyPal can send personalized reminders to patients to wear their devices and charge them as needed. These reminders can be customized based on the patient’s schedule and preferences, ensuring they are timely and effective.
User-Friendly Interface: The platform offers a simple, intuitive interface for both patients and clinicians. This reduces the complexity and enhances the user experience, encouraging consistent use of wearables.
Engagement and Education: Clinical StudyPal includes features to educate patients about their devices and the importance of consistent wear for their health and the success of the clinical trial. This educational component can significantly improve compliance rates.
Real-Time Monitoring and Feedback: The platform provides real-time monitoring of wear compliance, allowing clinical teams to intervene promptly if a patient is not wearing their device as required. Immediate feedback can also be provided to patients to reinforce positive behaviors.
Privacy and Security: Clinical StudyPal ensures the highest standards of data privacy and security, addressing one of the primary concerns patients have regarding wearables. This transparency and assurance can increase patient trust and compliance.
Integration and Flexibility: The platform is designed to be compatible with a wide range of wearable devices and can be integrated into various clinical trial protocols. This flexibility ensures that Clinical StudyPal can be tailored to meet the specific needs of each study and patient population.
Patient-Centric Approach: By prioritizing the patient’s comfort and convenience, Clinical StudyPal encourages greater compliance. Features like adjustable wearing schedules and personalized device settings ensure that the wearables fit seamlessly into patients’ lives.
Conclusion
Wearable devices present a powerful tool for remote monitoring in healthcare and clinical trials. However, the potential of these technologies can only be fully realized when wear compliance issues are effectively addressed. Clinical StudyPal offers a comprehensive solution to manage these challenges remotely, ensuring high-quality data collection and fostering better patient outcomes. By prioritizing patient engagement, comfort, and education, Clinical StudyPal is setting a new standard for wearable compliance in the digital health era.